When Drew Weissman and Katalin Karikó got their COVID-19 vaccine jabs before Christmas at the University of Pennsylvania, the cameras were rolling.
Three decades of persistence and painstaking research into mRNA vaccine science had paid off.
They’d proved the sceptics and naysayers wrong. What started out as a wild idea was suddenly a world-changing, lifesaving technology.
“We were incredibly excited when we saw the results of the Phase 3 trial that our vaccine worked, it was safe, and had 95 percent efficacy,” Professor Weissman said.
The COVID-19 vaccines developed at unprecedented speed by Pfizer and Moderna use technology licensed from the University of Pennsylvania team.
Professor Karikó is now also a senior vice-president of BioNTech, the German company behind the Pfizer vaccine.
But Professor Weissman isn’t resting on his laurels.
“My family always yells at me because I’m not excited enough, and they’re right,” he said.
“I’m already moved on to the next thing, the next vaccine, the next gene therapy.”
Space to play or pause, M to mute, left and right arrows to seek, up and down arrows for volume.
The promise of mRNA vaccines and therapeutics for other diseases could be explosive.
“We have five clinical trials set up ready to do, and they’ve been delayed [by] the coronavirus,” Professor Weissman said.
“We’ve probably made 30 different vaccines against different pathogens.
“Influenza, HIV, malaria, norovirus … a whole bunch.
“In just about every animal model, we had 100 percent efficacy. The vaccine is incredibly potent, and it gives very strong protective responses.”
But some scientists believe Australia is at risk of missing out on the full fruits of mRNA vaccine technology due to lack of investment and foresight.
Australia’s vaccine manufacturing deal
The Federal Government plans to start distributing COVID-19 vaccines at the end of the month, initially prioritising high-risk groups.
It leapt out of the gate early in the pandemic to secure doses of Pfizer’s mRNA vaccine, the first to be approved by our drug regulator, the Therapeutic Goods Administration.
Last week, an additional 10 million doses were procured, taking the total to 20 million.
But to meet ongoing demand, Australia is backing a different horse with local manufacturing.
Australian biotech giant CSL is only licensed to make the Oxford-AstraZeneca vaccine, a DNA-based vaccine which doesn’t need to be stored at -70 degrees Celsius, unlike mRNA vaccines.
It’s proved effective against COVID-19 in clinical trials, but less so than the mRNA vaccines — 62 per cent to 90 per cent effective (depending on dosing) versus Moderna and Pfizer’s 94 per cent and 95 per cent respectively.
However, CSL has not announced any plans to make mRNA vaccines in the future.
“Part of it is commercial decision-making,” said Colin Pouton, professor of pharmaceutical biology at Monash University.
“Obviously they’ve already agreed and are making deals with AstraZeneca’s vaccine, and that’s going to be a big effort for them.”
Calls to make more vaccines in Australia
But there are mounting calls for Australia to manufacture mRNA vaccines as a matter of vaccine security and savvy investment.
“It’s just the sort of high-tech industry that Australia is well suited to get into,” Professor Pouton said.
“We have a very good scientific base, very good technical experience and we have the sort of infrastructure already which fits well with this sort of technology.”
Importantly, it might also help pave a faster path out of this pandemic.
Like the rest of the world, we are wholly reliant on just two pharmaceutical companies, with operations in the US and Europe, to meet demand.
“We are at their mercy. And that that’s not an ideal situation,” said Archa Fox, an RNA biologist at the University of Western Australia.
Battles over shortfalls have already erupted, with the European Union introducing export controls to shore up its own supplies.
In late October, Federal Minister for Industry, Science, and Technology Karen Andrews said CSL (which makes our seasonal flu jabs) could make protein-based vaccines relatively quickly.
Determining the facilities needed to manufacture mRNA vaccines would “take significantly longer to do, but the work is underway,” she said.
“I would hope that we would be able to do it in about the nine-month to 12-month time frame.”
Professor Pouton believes it could happen quicker.
“I don’t actually agree with that assessment,” he said.
“Australia is very capable of making an mRNA vaccine … we can utilise facilities that we already have.”
Ms Andrews said an audit identified “some companies in Australia with mRNA production capability.
“We are currently working with them to explore if that capability could be scaled up into the future,” she said.
But the Government is yet to confirm any details on funding, logistics or a timeline for action.
Its Mid-Year Economic and Fiscal Outlook (MYEFO) statement in December mentioned funding to “develop a business case to inform future investments in onshore mRNA platform-based vaccine manufacturing capability and capacity”.
Again, no dollars were attached, citing “commercial sensitivities”.
“Producing mRNA vaccines at scale is a brand new challenge the entire world is grappling with and will take time to develop,” Ms Andrews said.
Professor Pouton said scaling up production was feasible because of the low amounts of mRNA required by the Pfizer and Moderna vaccines — 30 micrograms and 100 micrograms per dose, respectively.
“30 micrograms means that if you make 30 grams [of mRNA], you have a million doses,” he said.
What would it take to make mRNA vaccines?
Professor Pouton’s team are working on two mRNA vaccines for COVID-19 in collaboration with the Doherty Institute in Melbourne.
“Interestingly, it’s not actually that difficult,” he said.
“Making the Oxford-AstraZeneca vaccine is actually much more challenging from a pharmaceutical point of view.”
Until COVID-19, mRNA vaccines had never been tested in large-scale clinical trials before or approved for use in humans.
“It’s the new kid on the block with vaccine technology,” Dr Fox said.
“I don’t think anybody could have predicted just how effective these vaccines were.
“Sometimes it is hard to break through with a new technology. But this really has proved the naysayers wrong.”
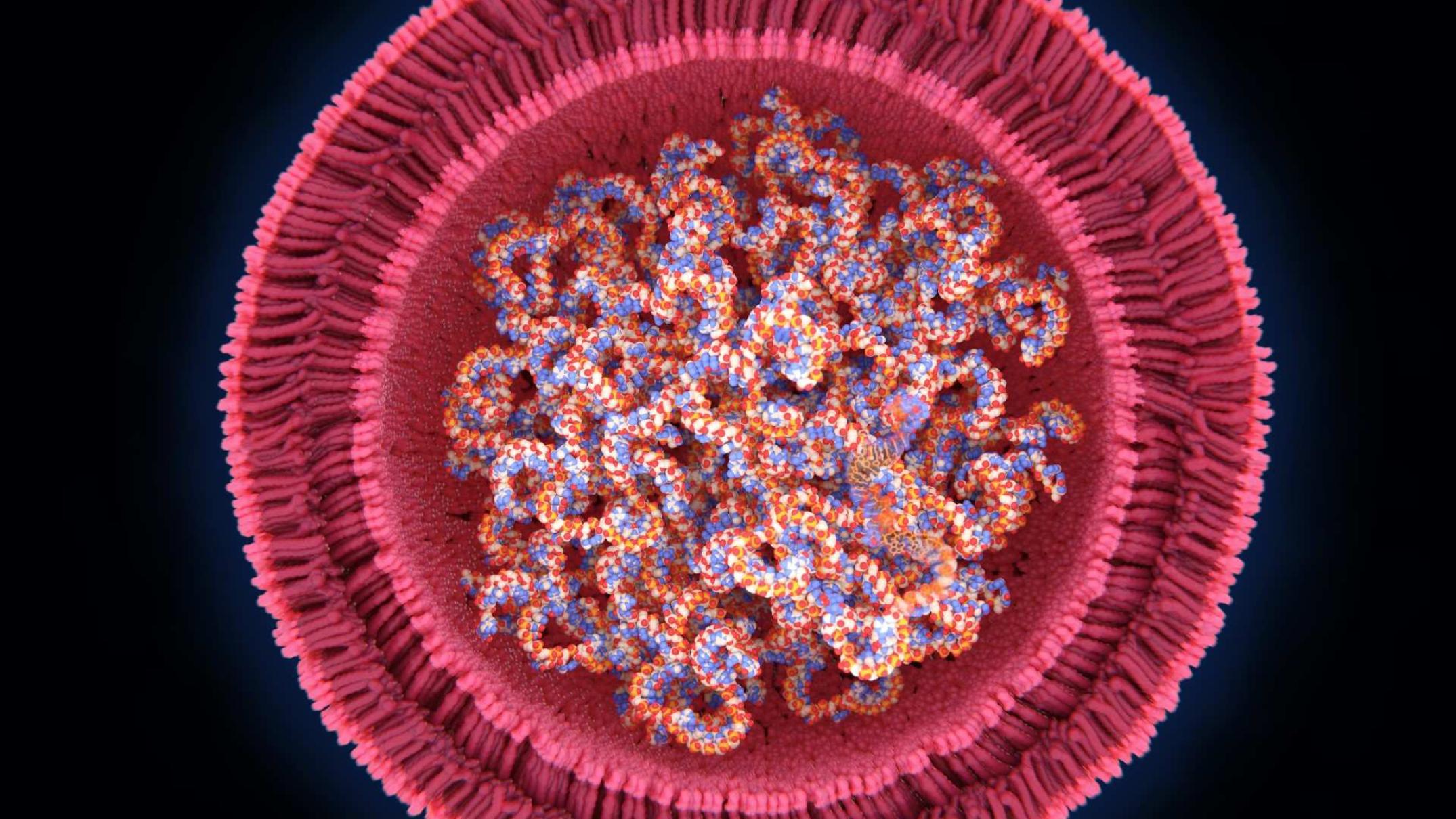
mRNA, or messenger RNA vaccines, borrow a mechanism that all of your cells use to make all the proteins in your body. It helps translate the message in your DNA’s genetic code into proteins.
mRNA vaccines work by injecting a particular molecule of mRNA into your cells that makes the spike protein found on the surface of the SARS-CoV2 virus.
This trains your immune system to “remember” the virus spike and make antibodies against it, so that if it ever meets the real virus, it ready to pounce and mount an immune response.
“Instead of using the old-fashioned vaccines where they … use a deactivated virus or sometimes even an attenuated virus, with all the other complexities that involves, here you’re using a very clean presentation of a single protein,” Professor Pouton said.
A few main steps are involved in making mRNA vaccines. Simply put, they include:
- synthesising DNA by growing it in E. coli bacteria
- using the DNA and various ingredients to make mRNA in a bioreactor
- purifying and encapsulating the mRNA in fatty (lipid) nanoparticles to help it enter your cells without degrading
- processing and packaging the vaccine into vials
“Once the RNA is made, the vaccine is essentially done,” vaccine inventor Professor Weissman said.
Professor Pouton believes most of this can already done by Australian scientists and facilities.
“Not necessarily in one location, but [with] a bit of teamwork and cooperation, it can be done,” he said.
“The step that they’re less familiar with is taking the DNA code and producing mRNA from it.
“But it’s actually a very simple thing to do once you’ve got the ingredients. A one pot, if you like, a test tube process.
“It doesn’t require a human or mammalian cells or anything like that, it’s just mechanics involved here. It’s not something that requires a massive plant.”
Other governments leading the way
Legal agreements over licensing the technology take time to negotiate, however.
“We’re not alone in having this inability to have the sovereign manufacturing of RNA vaccines,” Dr Fox said.
Last week, French President Emmanuel Macron confirmed his country had negotiated with Moderna and Pfizer to enable local production in French labs.
The Thai Government has also made headway.
“I’ve been working with Thailand for years on making RNA vaccines for diseases that were Thailand-specific,” Professor Weissman said.
“[They] made the really correct decision that if the [COVID-19] vaccine was made in the West, it would be years before Thailand, the rest of South-East Asia, Africa and other [developing] countries would ever get access.”
The Thai Government will fund a manufacturing facility to help supply vaccines to their own population and surrounding low-moderate-income countries.
“To them, it was obvious that RNA was the way to go,” Professor Weissman said.
But what will convince Australian companies to put their oar in something so new?
“They see it as a risk — will this investment that they make be useful to them in the future?” Professor Pouton said.
Initial government investment would help kickstart industry, he added.
“We have people who understand good manufacturing practice for human use, and … the technology once they get their head around it. It’s not as difficult as some of the things that they’re already doing,” Professor Pouton said.
More where that came from
mRNA vaccines have had a baptism of fire during the COVID-19 pandemic, and their impressive clinical trial results have surprised even their inventor.
“Anybody who does research knows that when you work in mice and even in monkeys, the results are always or usually much better than what you see in people,” Professor Weissman said.
“I was relieved and just incredibly happy.”
But it’s what’s waiting in the wings that is turning Australia’s small community of RNA biologists into lobbyists.
“We know that we can make it at the scale that is required … there is this huge untapped potential for all sorts of future applications. It is definitely a game changer,” Dr Fox said.
She points to local scientists doing frontier work in mRNA therapeutics — ranging from cancer immunotherapies including for childhood cancers, therapeutic vaccines, protein replacement therapies, regenerative medicine and other infectious disease vaccines.
“I hope … we take a forward-looking approach and think about future pandemics or even developing vaccines against the variants,” Dr Fox said.
“It makes a lot of sense to get this up and running in Australia.”
To her, COVID is a prime example of how fundamental research over many years can be pivotal when we least expect it.
mRNA vaccines are unlikely to be a one-pandemic wonder.